
Results from a prespecified interim analysis showed that. Eligible patients in the control arm of the study who received docetaxel will be allowed to continue treatment or cross over to the group receiving opdivo.
The american cancer society estimates that for 2018 about 234,030 new cases of lung cancer will be.
Opdivo lung cancer survival. Duration of response, or the length of time a tumor continues to respond to treatment without growth or spread, was 10.2 months for the opdivo plus yervoy group, 11.2 months for the opdivo alone group and 8.1 months for the. To date, opdivo has shown improved efficacy in the neoadjuvant or adjuvant treatment of four tumor types: Eligible patients in the control arm of the study who received docetaxel will be allowed to continue treatment or cross over to the group receiving opdivo.
Lung cancer, bladder cancer, esophageal/gastroesophageal junction cancer and melanoma. Bms’ latest findings show that it wants to remain competitive if not best its rival merck’s keytruda. Opdivo shows promise against lung cancer.
Bristol myers’ opdivo delivers increased survival in nsclc. One trial found that people who took opdivo had longer survival compared. The american cancer society estimates that for 2018 about 234,030 new cases of lung cancer will be.
Opdivo, alone or combined with yervoy, extends lung cancer patients’ survival, phase 1/2 trial shows. November 2, 2017 • by liz highleyman Lung cancer is the second most common cancer in both men and women and accounts for about 14% of all new cancers.
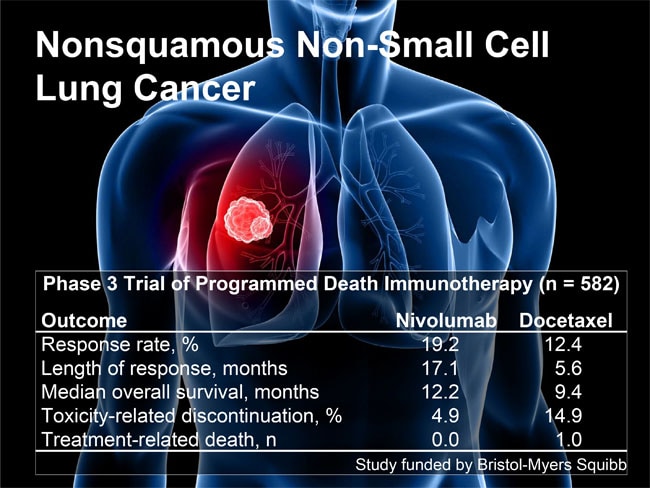
March 16, 2018 rr hem/onc updates. Nivolumab versus docetaxel in patients with previously. 40% at 2 years vs chemotherapy:
Opdivo (nivolumab) has shown promise as a treatment for recurring small lung cancer patients who have a lot of genetic errors in their tumors, according to a phase 1/2 clinical trial. Long term survival outcomes with opdivo® in advanced nsclc. Updated pooled analyses of the checkmate 017 and 057 clinical trials comparing opdivo with taxotere was presented at the international association for the study of lung cancer world conference on lung cancer in september 2019 and showed that 13.4% of opdivo treated patients survived 5 years compared with only 2.6% of patients treated with taxotere.
Results from a prespecified interim analysis showed that. P=0.0015]), based on a prespecified interim analysis. Median duration of overall survival.
Opdivo outperformed the cancer drug taxotere in clinical trials of advanced and earlier treated lung cancer, with survival rates about double those seen with the older treatment after three years. 1 the median os was 12.2 months in the opdivo arm (95% ci:.