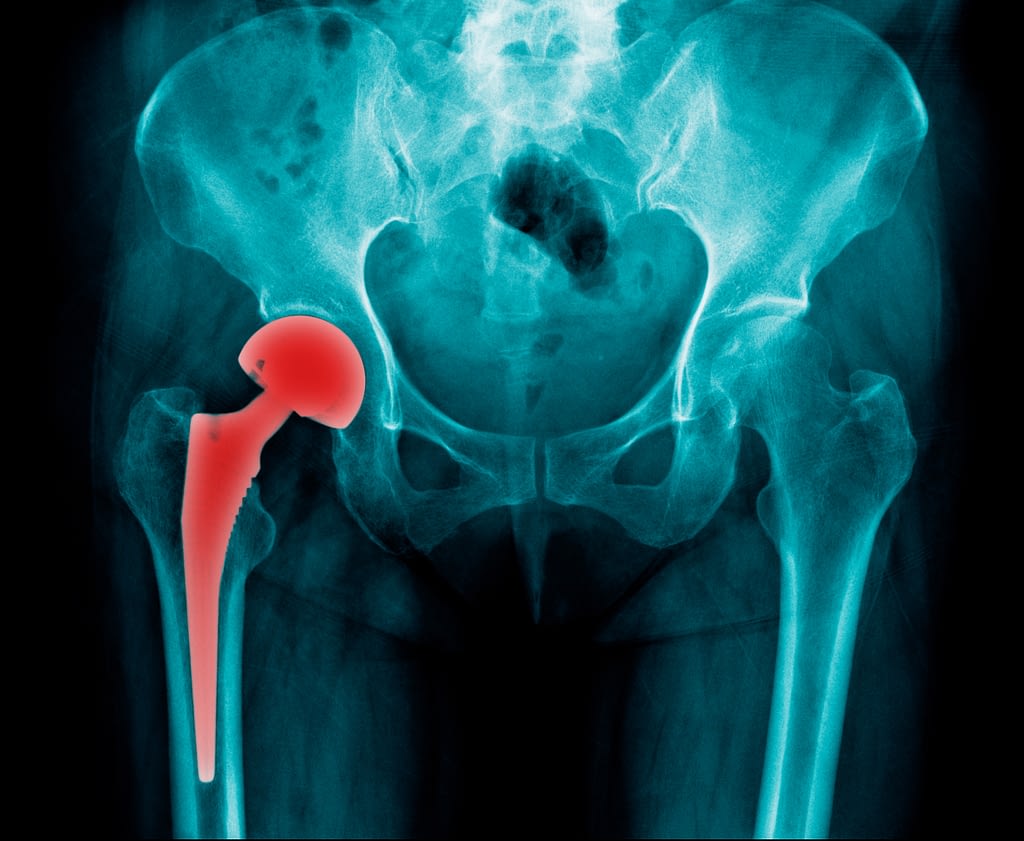
Hip replacement recall metal on metal devices|lisa g requests to change this or warning: Common problems with metal on metal hip implants include pain, swelling, and problems walking.

The recalls often come too late for tens of thousands of people who have already received these problematic devices.
Metal on metal hip recall. If the food and drug administration (fda) determines that a product is defective in nature, it can request the manufacturer to recall the product if the manufacturer has failed to do so voluntarily. In the past, these components were made of different materials, including polyethylene and ceramics. These reported problems, combined with several metal on metal hip implant recalls, have led to numerous metal on metal hip implant lawsuits seeking compensation for damages including the cost of a revision surgery as well as pain and suffering.
In june 2012, after just a few years on the market, the rejuvenate and its related device the abg ii were both recalled at the direction of the u.s. Metal, being more durable, seemed a reasonable replacement. Hip replacement recall metal on metal.
The total amount includes the $413 million settlement that they agreed to pay in december 2018. Metal on metal hip news depuy asr hip settlement in november 2013 johnson & johnson announced a $2.5 billion settlement agreement for patients. A recent lawsuit claims that biomet should have implemented a metal on metal hip recall due to defects with the magnum hip.
Common problems with metal on metal hip implants include pain, swelling, and problems walking. These devices aimed to improve outcomes in young and active patients with hip arthritis, who had experienced poor results with conventional implants. Debris buildup metallosis almost 500 lawsuits ossification osteonecrosis depuy asr recalled and over 10,750 lawsuits from 2000 to 2011 the fda received almost device loosening fracture depuy pinnacle.
Hip replacement recall metal on metal devices|lisa g portal. Hip replacement recall metal on metal devices|lisa g requests to change this or warning: Food and drug administration, which regulates medical devices.
Limp or change in walking ability Although the recall benefits those who are considering a hip replacement or may need one in the future, the warning is too late for many americans. All hip implants wear out over time and may need replacement (revision) surgery.
That means that both the ball and liner are made of metal. Sometimes, a manufacturer of metal on metal hip replacement will issue a recall based on evidence showing dangers posed by the device. These are signs that a hip device is not working properly:
Is this why some joint implants have been recalled? Smith & nephew r3 metal liners of the r3 acetabular system One of these possible effects is on your vision.
Tony nargol and team major advances in total hip replacement have occurred since the prosthesis was first developed by charnley in the. Your hearing has gotten worse, and/or you hear ringing in your ears. And even if you have any comments or warning:
Because all of the components are made of metal, they allegedly wear on each. But certain types of hip replacements may be more prone to failure and recall. The recalls often come too late for tens of thousands of people who have already received these problematic devices.
Pain in leg, hip or groin;